Article Generator
Interactive, Evidence-Based Review and Article Creation Workflow
Overview
The Article Generator feature is designed to automate and streamline the creation of comprehensive scientific reviews and analytical articles on complex medical topics.
Users start by describing their topic or task (e.g., “Review the survival benefit of ribociclib compared to other CDK4/6 inhibitors in HR-positive, HER2-negative advanced breast cancer”), and the system systematically guides them through technical brief creation, outline development, and full article drafting, with feedback and approval at every step.
How It Works
- Initial Input:
User submits a short topic description or task objective in plain English (or any supported language).- Example:
“Ribociclib - survival benefit in HR-positive, HER2-negative advanced breast cancer. Review the evidence of ribociclib benefits over competitors.”
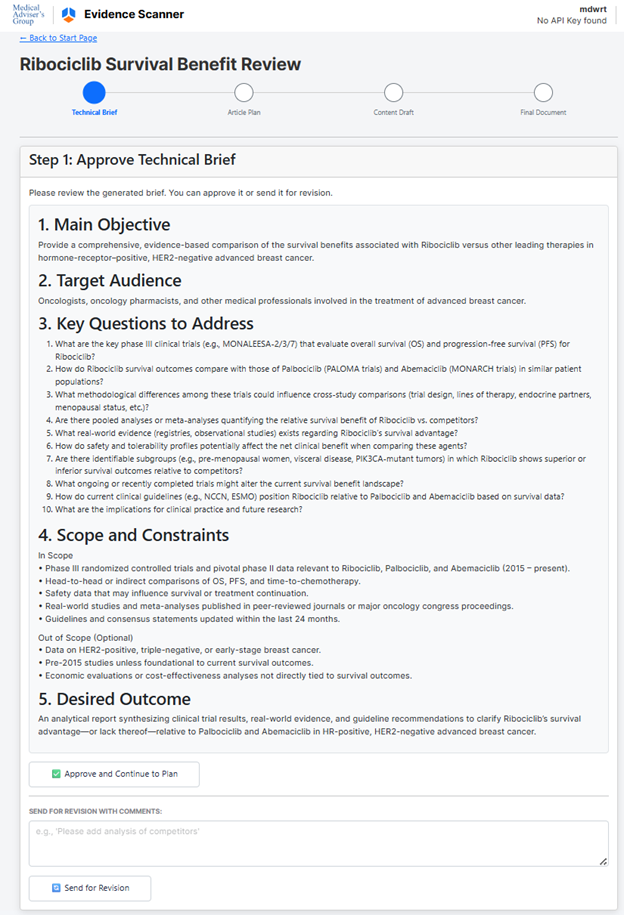
- Technical Brief Generation:
- The system analyzes the input and automatically drafts a technical brief covering:
- Main objective
- Target audience
- Key questions to address
- Scope and constraints (what to include/exclude)
- Desired outcome (target format, length, structure)
- The system analyzes the input and automatically drafts a technical brief covering:
› Outline Example
Main Objective
Produce an evidence-based review that evaluates the survival benefit of ribociclib in hormone-receptor–positive, HER2-negative advanced breast cancer and compares these outcomes with those of competing CDK4/6 inhibitors.
Target Audience
- Oncologists and oncology pharmacists
- Clinical researchers and guideline developers
- Healthcare decision-makers (e.g., formulary committees, payers)
Key Questions to Address
- What are the pivotal clinical trials investigating ribociclib in HR-positive, HER2-negative advanced breast cancer (e.g., MONALEESA-2, -3, -7), and what survival endpoints (overall survival, progression-free survival) were reported?
- How do ribociclib’s survival outcomes compare quantitatively with those of other CDK4/6 inhibitors (palbociclib, abemaciclib) across similar patient populations and lines of therapy?
- What subgroup analyses (age, menopausal status, visceral disease, prior therapy) demonstrate differential survival benefit with ribociclib versus competitors?
- What are the safety and tolerability profiles of ribociclib relative to its competitors, and how might these influence overall clinical benefit?
- How do indirect comparison methods (e.g., network meta-analyses or matching-adjusted indirect comparisons) further clarify ribociclib’s comparative survival advantage?
- What ongoing or recently completed trials could modify the current understanding of ribociclib’s survival benefit?
- What clinical practice guidelines or real-world evidence support or challenge ribociclib’s perceived survival advantage?
- Scope and Constraints
In Scope
• Phase II/III randomized controlled trials and pooled analyses of ribociclib, palbociclib, and abemaciclib in HR-positive, HER2-negative advanced/metastatic breast cancer
• Overall survival (OS), progression-free survival (PFS), and landmark survival rates (e.g., 3-year OS)
• Safety data directly affecting benefit-risk assessment (e.g., neutropenia, QT prolongation)
• Subgroup and biomarker analyses relevant to survival outcomes
• Indirect comparison techniques when head-to-head data are lacking
• Publications, congress abstracts, and regulatory reviews from 2016 to present
Out of Scope
• Early-stage breast cancer data
• Non-CDK4/6 targeted therapies unless used as control arms
• Trial data published before 2016 (pre-CDK4/6 era)
• Cost-effectiveness or pharmacoeconomic analyses
Desired Outcome
An analytical report (~2,500-3,000 words) with the following structure:
- Executive summary of survival findings
- Methods (literature search and comparison approach)
- Detailed results for ribociclib trials
- Head-to-head or indirect comparisons with palbociclib and abemaciclib
- Safety and tolerability comparison
- Discussion of clinical implications and gaps
- References (primary literature, guidelines, regulatory documents)
User Review:
The user can approve the brief or leave free-form comments for revision. The system will update the brief according to feedback until approved.
- Article Outline Development:
- Once the brief is approved, the system proposes a detailed outline (plan),
› Detailed outline example
Executive Summary
Task: Synthesize the key survival findings for ribociclib versus palbociclib and abemaciclib, highlighting absolute and relative improvements in overall survival (OS) and progression-free survival (PFS) that are most relevant for oncologists and decision-makers.
Methods
Task: Describe the systematic literature search (databases, congresses, cut-off dates), inclusion/exclusion criteria, data extraction process, and the statistical approaches used for direct and indirect comparisons.
Ribociclib Clinical Trial Results
Task: Present detailed efficacy outcomes (OS, PFS, landmark rates) from MONALEESA-2, ‑3, and ‑7, including key subgroup analyses (age, menopausal status, visceral disease, prior therapy) and relevant safety data.
Comparative Survival Outcomes with Other CDK4/6 Inhibitors
Task: Quantitatively compare ribociclib’s survival endpoints with those reported for palbociclib (PALOMA trials) and abemaciclib (MONARCH trials) in similar clinical settings and lines of therapy.
Subgroup and Biomarker Analyses
Task: Evaluate differential survival benefits across predefined subgroups and emerging biomarkers, contrasting ribociclib with palbociclib and abemaciclib where data permit.
Safety and Tolerability Comparison
Task: Summarize and contrast adverse-event profiles (e.g., neutropenia, diarrhea, QT prolongation) and discuss how safety considerations influence overall clinical benefit–risk assessments.
Indirect and Network Meta-Analysis Evidence
Task: Review published network meta-analyses or matching-adjusted indirect comparisons that contextualize ribociclib’s comparative survival advantage in the absence of head-to-head trials.
Emerging Data and Ongoing Trials
Task: Outline ongoing or recently completed trials that may refine current understanding of ribociclib’s survival benefit and note anticipated readouts.
Clinical Practice Guidelines and Real-World Evidence
Task: Summarize guideline recommendations and real-world survival data that either support or challenge the superiority of ribociclib.
Discussion, Limitations, and Future Directions
Task: Interpret the collective evidence, address methodological limitations, highlight gaps in knowledge, and propose future research priorities.
References
Task: Compile primary trial publications, meta-analyses, guideline documents, and regulatory reviews cited throughout the report.
Section-by-Section Drafting:
Finalization:
Here is an example:
Ribociclib Survival Benefit Review_article_250726__1745
Key Features & Workflow Advantages
- Step-by-step, interactive workflow:
Users can review and comment at each stage (brief, outline, every section), ensuring the final product fully meets their needs. - Evidence-driven:
The system uses a combination of web search and user-uploaded documents, guaranteeing high-quality, up-to-date, and referenced content. - Transparency and control:
At each step, users see the rationale and sources, and can direct changes before moving forward. - Automated referencing:
All references are formatted with hyperlinks; citation numbering is consistent throughout. - Versatile output:
Ideal for evidence reviews, literature summaries, scientific white papers, competitive analyses, or congress materials.
Core Benefits
- Collaborative drafting:
The system acts as an AI “co-author” but always keeps the user in the driver’s seat. - Eliminates blank-page syndrome:
Even the most complex reviews are broken down into manageable, reviewable steps. - Flexible sourcing:
Combines web-based evidence with proprietary or internal documents as needed. - Regulatory ready:
Fully referenced, transparent, and editable output, compatible with medical and scientific standards.
Summary
From a simple task description to a ready-to-publish scientific review, the Article Generator leads users through a transparent, interactive process, allowing full control, commenting, and approval at every stage — with output delivered in Microsoft Word Online format.